The history of laboratory-grown diamonds
First attempts

Image courtesy Library of Congress Prints and Photographs Division; Washington, D.C., 20540, USA; http://loc.gov/pictures/resource/ggbain.02112
In 1867, a young boy named Erasmus Jacobs discovered the 20-carat ‘Eureka Diamond’ on the banks of the River Orange in South Africa. This marked the beginning of the South African diamond rush. Geologists found the first examples of diamonds in the nation’s native rock, kimberlite, which forms deep within the earth. Scientists began to surmise heat and pressure were key to its formation—hence, the beginning of high-pressure, high-temperature (HPHT) efforts to synthesize diamond.
1878: An explosive start
Scottish chemist James Ballantyne Hannay was among the first to attempt to produce diamonds in a laboratory setting. He was also the first to claim success, albeit prematurely, and is regarded as a piece of scientific mythology.
Hannay had a rather dangerous way of approaching diamond synthesis. He heated a mixture of carbon-rich materials and lithium to a red heat in sealed gun barrels. His theory was the heat and pressure would force the contents inside to crush itself into diamonds. This was hazardous, as the tubes tended to violently explode. While his techniques were eventually determined to be conducive to diamond formation, it required capabilities not available in Hannay’s time.
Nonetheless, the samples he brought to the public were certainly diamonds, and, in 1880, the Royal Society published Hannay’s paper on the synthesis of diamond. At the time of his discovery, it is said Hannay was offered a very large sum if he would drop the whole thing; it is also said the Stock Exchange and the Amsterdam Diamond Bourse were distinctly ‘het-up’ by the report.1
Ultimately, however, scientists in 1975 ultimately proved Hannay’s creations were actually natural diamonds and not made from his experiment at all. The minute particles he claimed to have produced were handed over to the Mineral Department of the British Museum, where they remain to this day.
Hannay’s methods were extremely violent and eventually had an impact on his health:
“The continued strain on the nerves, watching the temperature of the furnace, and in a state of tension in case of an explosion, induces a nervous state which is extremely weakening, and when the explosion occurs it sometimes shakes one so severely that sickness supervenes.”2
1889: High-temperature headway
Inspired by the discovery of diamonds at the Canyon Diablo meteorite site in Arizona (1891), Nobel-prize winning French chemist and pharmacist Ferdinand Frédéric-Henri Moissan paved the way for high-temperature research in 1889 (Figure 2).
Based on his observations of findings in the meteorite, Moissan theorized diamonds could be synthesized by crystallizing carbon under pressure from molten iron. He even developed an electric-arc furnace to enable high-temperature research. (Moissan’s discovery of the importance of iron in dissolving carbon was essential to the eventual success achieved by H. Tracy Hall in 1955.)
While it is true tiny octahedral diamond crystals were extracted following one of Moissan’s experiments, it was later determined they were planted there by a sympathetic lab assistant who hated seeing his boss fail—talk about employee dedication!
Fear not, though: Moissan did grow crystals in his experiments. The jewellery industry can thank him, for—you guessed it—synthetic moissanite. The popular diamond simulant is named after him.

Harvard University, courtesy AIP Emilio Segrè Visual Archives
Many other scientists replicated Moissan’s experiments, but no one created diamond. One critical ingredient was missing: pressure. It was not until 1935 when additional significant discoveries were made. This had a lot to do with evolving technology and industrialization, as well as the race to win World War II.
1935: The dawn of high-pressure research
Percy Williams Bridgman was an American experimental physicist who made major contributions with his high-pressure research. This served as the basis to the first successful synthesis of diamonds.
Bridgman worked on high-pressure experiments, including developing a special press able to apply high pressure (the Bridgman anvil, Figure 3) and a special type of self-sealing gasket (the Bridgman seal), which enabled contents under pressure to not leak out of the chamber. He received the Nobel Prize for Physics in 1946 for his contributions to high-pressure research.
While Bridgman did not synthesize diamonds (despite repeated attempts), his experimentation led directly to their eventual synthesis by scientists of the General Electric Company (GE) in 1955. Without his findings, work at very high pressures would not have been possible.
In the meantime, researchers were also better understanding the behaviour of carbon, including how it performed at certain pressures and temperatures (i.e. when it was stable as graphite and when it was stable as diamond).
Early determinations of the carbon phase diagram by Frederick D. Rossini and Ralph S. Jessup in 1938 (refined in subsequent years) revealed diamond is the higher-pressure, lower-temperature form of carbon, likely forming at a depth greater than 100 km.
This related to the concept of the geothermal gradient, which is the increasing temperature with respect to the increasing depth beneath the surface of the earth (Figure 4). This helped move forward the importance of pressure in creating diamond in a laboratory.
By the 1940s, WWII jeopardized the world’s supply channels of natural diamonds (which were essential for industrial cutting tools) and the race was on to grow diamonds in a laboratory. The increase in the use of hard-metal tools in the munitions industry required industrial diamonds. For many, in fact, it was believed whoever could solve the diamond supply problem would win the war.
In the United States, GE launched ‘Project Superpressure’ to research the process of growing diamonds. Meanwhile, scientists in Sweden were working on a diamond-making project called ‘QUINTUS’ at ASEA (Allmanna Svenska Elektrika Aktiebolaget)—also known as the Swedish General Electric Company.
At this time, here is what scientists already knew:
- Diamonds are made of carbon.
- Under certain conditions, graphite can turn into single-crystal diamond.
- Natural diamonds are found in extinct volcanic pipes and brought to the surface in lava. They form at depths of around 200 km within the earth where pressures and temperatures are very high.
- Diamonds formed in a meteor crater indicate high pressures (suggested by the meteor impact); diamond crystals surrounded by metals suggest the need for a metallic flux to dissolve carbon so carbon atoms will re-form into diamond.
- Graphite, the diamond allotrope, is extremely stable and resistant to change. Even at very high pressures and temperatures, the carbon atoms do not break apart and re-form into diamond. Thus, to create diamonds, scientists would have to find a way to sustain very high temperature and very high pressure simultaneously (Figure 5).
Between diamond and graphite (i.e. carbon allotropes), diamond is the most stable configuration of pure carbon at high pressures. As such, most diamond synthesis efforts were focused on HPHT growth methods.
Concurrently in the 1950s, however, research started on the growth of diamond at metastable conditions (i.e. at low pressures). This research started in the Soviet Union and in the United States by experimenting with the growth of diamond by pyrolysis (decomposition caused by high temperatures) of hydrocarbon gases at the relatively low temperature of 800 C (1472 F). Ultimately, this low-pressure process became known as chemical vapour deposition (CVD).
1952: First success
The first successful, reproductive diamond synthesis (by any method) was achieved by William G. Eversole of the Union Carbide Corporation between 1952 and 1953 (Figure 6). He accomplished the feat using low-pressure CVD—not HPHT growth. Eversole succeeded in depositing diamond on a diamond substrate from carbon monoxide and low molecular weight hydrocarbons. His work was foundational to CVD research.
So, why isn’t Eversole’s achievement recognized, officially, as the first laboratory-grown diamond?
Most notably, he did not report on his results until a decade later in 1962. Additionally, while Eversole successfully produced diamond, low-pressure synthesis was plagued by extremely slow growth rates of no greater than 0.1 micrometres (ɥm) per hour. The process was also viewed with some skepticism in terms of its commercial viability. Later, both American and Russian teams had success in creating diamond films on diamond and non-diamond materials. This led to massive research and developments on diamond coatings and diamond-like carbon.
Thus, despite this early start, diamond synthesis using low-pressure methods like CVD only began to generate significant scientific and economic interest in the 1980s. Instead, focus largely remained on high-pressure, high-temperature synthesis of diamond.
With the focus on HPHT, however, suddenly two major breakthroughs occurred within a year of one another by two independent teams on different continents.
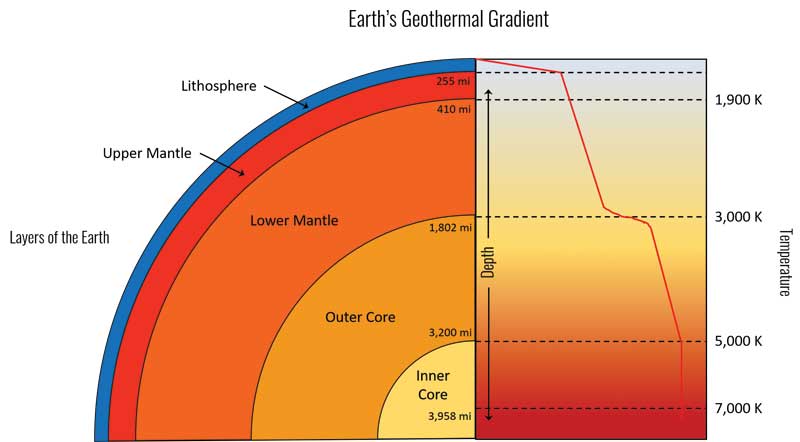
Image by Alethea Inns/GS



