The acid test: Preserving pearls’ fragile beauty
By Elisabeth Strack

Pearls have been admired and cherished since ancient times. They were highly appreciated in Greek and Roman antiquity, and ever since, they have enjoyed a positive reputation as objects of adornment and value. Kokichi Mikimoto’s vision that one day every woman should wear and own a pearl necklace has nearly come true.
Images © Elisabeth Strack
Pearls are also fascinating jewellery objects—they distinguish themselves from most gemstones because they grow inside of molluscs and are therefore considered products of biomineralization. This process involves an organic substance called conchioline, which is usually described as the ‘organic matrix.’ It is a protein of the keratin type that serves as a type of mortar for the calcite prisms and aragonite platelets of which the pearl is mainly composed.
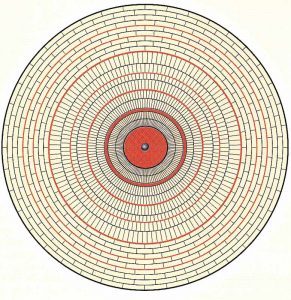
Most natural pearls have a rather large inner core made up of concentrically arranged calcite prisms. This is followed by an outer layer of aragonite platelets in a similar concentric arrangement, which gives rise to the brick-type structure usually described as nacre (at right). The chemical composition of both calcite and aragonite is calcium carbonate (CaCO3), but they have different crystal systems (trigonal for calcite and rhombic for aragonite).
Due to their chemical composition, pearls are highly sensitive to acids and have to be handled with care. The reason for this is acids trigger a decomposition process in which carbonic acid is set free, thus causing a pearl to effervesce. This leads to damage to the pearl’s surface that can go as far as complete dissolution. The latter can be achieved within minutes when a pearl is immersed in hydrochloric acid—it will disappear quickly in front of the observer’s eye.
Cultured pearls (such as akoya, South Sea, Tahitian, and freshwater cultured varieties) have a similar chemical composition to their natural counterparts. This means they will also react with acids, and have to be handled with the same care as natural pearls.
While it can be assumed pearls will not normally come into contact with acids in a jewellery store, attention has to be paid inside the workshop. Pearls always have to be kept at a safe distance from the strong acids a jeweller or goldsmith uses to determine the purity of gold, as they contain both hydrochloric and nitric acid. Special care has to be exercised when testing a piece of gold jewellery set with pearls.







